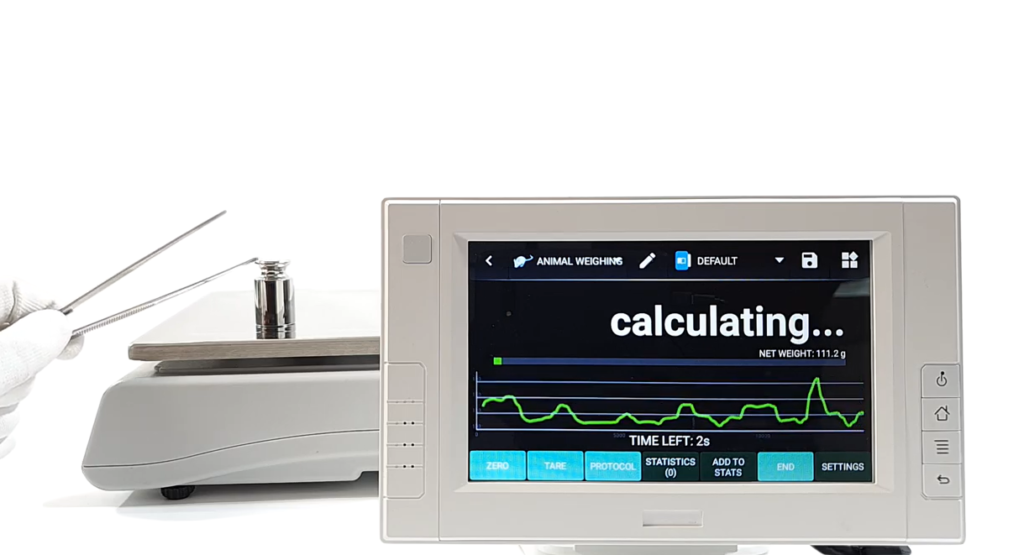
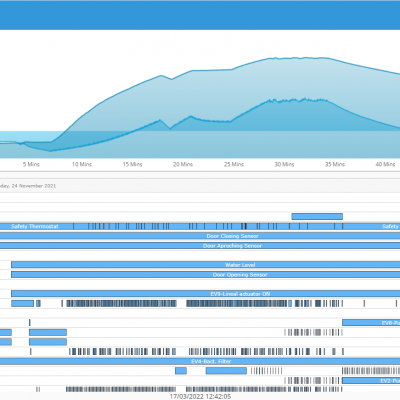
HMI solutions
Get HMI hardware, software and development services out of one hand.
Get cutting edge device management solutions for pharma and industrial devices.
On average 4 months development time and 70% cost savings due to use of ready-made components.